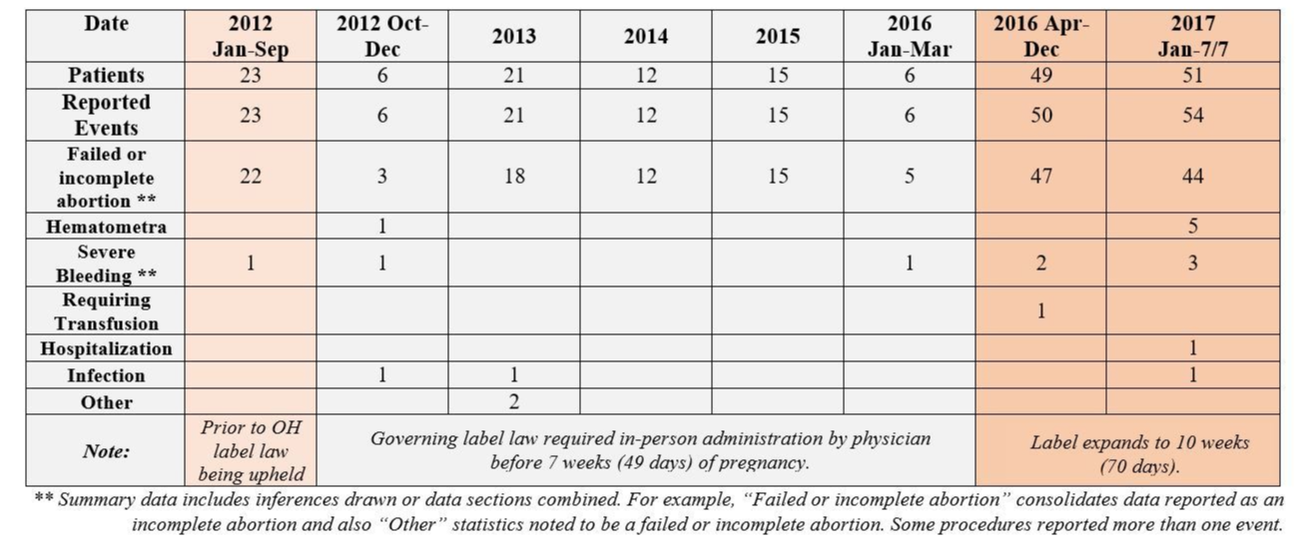
Background: On March 29, 2016, the FDA announced sweeping changes to the label of the abortion-inducing drug, RU-486. Changes to the label expanded the date in pregnancy for which the pill could be used (from 7 weeks to 10), altered the approved dosing rates, eliminated the requirement that only a physician administer the pill, and allowed the second dose of the regimen to be taken at home. The FDA did not remove the black box warning on the label, a note called the "Risk Evaluation and Mitigation Strategy" used for drugs with known potentially dangerous and serious side effects.
The move especially impacted states like Ohio, where state law requires that RU-486 be administered in person by a physician and exactly according to the label. Ohio's law was passed in 2004, but was not fully in effect until October of 2012 when it was upheld at the US 6th Circuit Court of Appeals.
The move to change the label was cheered by abortion advocates, but many pro-life and faith organizations expressed concerns that the Obama administration's move placed the lobbying efforts of abortion advocates before the interests of women's health and safety and the lives of unborn children.
At Greater Columbus Right to Life, we have undertaken an exhaustive review of the preliminary data available since the label change modified Ohio's prescribing parameters, and we have found that the incidence of serious complications related to RU-486 (mifeprex) has skyrocketed since April of 2016, supporting the claim that the FDA's decision prioritized the powerful abortion lobby over women's health and safety.
Our study also led us to discover a tangled web of business delinquencies, unpaid taxes, missed filings, expired licenses, failed reporting, and more among some abortion providers. We have notified the Ohio Secretary of State and the Ohio Attorney General of our findings and asked them to take enforcement actions against these violations to the fullest extent of their offices' authority. We have also asked that where they are not provided sufficient authority to take action to protect the health, safety, and well-being of women that they notify us of those deficiencies so that we can share with policy makers and advocates the provisions in Ohio law where our current statutory provisions lack teeth.
In addition to referring our complaints for enforcement, we are asking that the FDA review the label for RU-486, calling on lawmakers to close serious loopholes in the abortion law that place women at risk and shield questionable practices from the public eye, and educating the general public that the abortion pill is dangerous for women and deadly for unborn babies. You can join us by signing our petition, sharing the information, joining our team, or making a donation today.
The move especially impacted states like Ohio, where state law requires that RU-486 be administered in person by a physician and exactly according to the label. Ohio's law was passed in 2004, but was not fully in effect until October of 2012 when it was upheld at the US 6th Circuit Court of Appeals.
The move to change the label was cheered by abortion advocates, but many pro-life and faith organizations expressed concerns that the Obama administration's move placed the lobbying efforts of abortion advocates before the interests of women's health and safety and the lives of unborn children.
At Greater Columbus Right to Life, we have undertaken an exhaustive review of the preliminary data available since the label change modified Ohio's prescribing parameters, and we have found that the incidence of serious complications related to RU-486 (mifeprex) has skyrocketed since April of 2016, supporting the claim that the FDA's decision prioritized the powerful abortion lobby over women's health and safety.
Our study also led us to discover a tangled web of business delinquencies, unpaid taxes, missed filings, expired licenses, failed reporting, and more among some abortion providers. We have notified the Ohio Secretary of State and the Ohio Attorney General of our findings and asked them to take enforcement actions against these violations to the fullest extent of their offices' authority. We have also asked that where they are not provided sufficient authority to take action to protect the health, safety, and well-being of women that they notify us of those deficiencies so that we can share with policy makers and advocates the provisions in Ohio law where our current statutory provisions lack teeth.
In addition to referring our complaints for enforcement, we are asking that the FDA review the label for RU-486, calling on lawmakers to close serious loopholes in the abortion law that place women at risk and shield questionable practices from the public eye, and educating the general public that the abortion pill is dangerous for women and deadly for unborn babies. You can join us by signing our petition, sharing the information, joining our team, or making a donation today.
Share on Social Media |
REVIEW THE DATA
|
Note: 2016 and 2017 abortion statistics are not yet available to the public, so a comparison of the incident per dose cannot be made. However, between 2013 and 2015, there were about 812 medical abortions performed each year (855 in 2013 and 791 in both 2014 and 2015), according to Ohio Department of Health records. In order for the use of the abortion pill to keep pace with the rate of reported complications, use of the pill would have to climb to more than 4,000 doses, and even at that use, the rates of hematometra, hospitalization, and blood transfusion complications show a significantly dangerous impact on women.